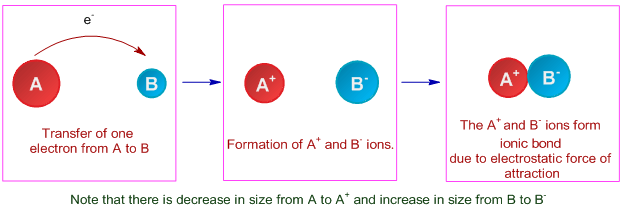
The ionic bond is the electrostatic force of attraction between two oppositely charged ions i.e., a positively charged cation and a negatively charged anion. It is formed due to complete transfer of electrons from one atom to another atom.
The steps involved in the formation of ionic bond can be summarized as:
a) An electropositive atom (metal) loses electron(s) to form a positively charged ion called as cation.
b) An electronegative atom accepts the electron(s) to form a negatively charged ion, otherwise known as anion.
c) Thus formed oppositely charged ions come closer to each other due to electrostatic force of attraction and get stability.
The formation of ionic bond between two atoms can be visualized as follows:

* An ionic bond is formed when the electronegativity difference between two bonding atoms is greater than 1.7 on Pauling's scale.
* It can be observed that an ionic bond is formed between an electropositive metal atom and an electronegative nonmetal atom.
KOSSELL'S ELECTRONIC THEORY OF CHEMICAL BONDING
The formation of ionic bond was explained by Kossell as follows:
* The atoms of inert gases are stable due to octet configuration (ns2np6) in their outer shell. Hence atoms must possess eight electrons in their outer shell to get more stability. This is referred to as octet rule.
* However, Helium is also highly stable due to 1s2 configuration.
* Therefore every atom tries to get nearest inert gas configuration either by losing or gaining electrons to form ionic bond.
* Only the electrons in the outer shell participate in bond formation. These electrons are also called as valence electrons.
The electrons in the inner shell are called as core electrons and do not participate in the bond formation.
Electrovalency: The number of valence electrons either lost or gained by an atom during the ionic bond formation is called electrovalency.
ILLUSTRATIONS
1) LiF (Lithium fluoride):
* In the formation of Lithium fluoride, the Lithium atom loses one electron to get nearest inert gas: Helium's configuration, 1s2.
* Whereas, the Fluorine atom gains one electron to get nearest inert gas: Neon's configuration, 1s22s22p6.
* Thus formed Li+ and F- ions get attracted towards each other to form the ionic compound LiF.
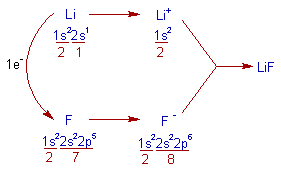
* The electrovalency of Lithium is one since it loses one electron.
* The electrovalency of Fluorine is also one since it gains one electron.
Note: The electronegativity difference between Li (1.0) and F (4.0) is 3.0, which is greater than 1.7. Hence they form ionic bond.
2) MgCl2 (Magnesium chloride):
* The Magnesium chloride is an ionic compound. The formation of ionic bond in this compound can be explained as follows.
* The Magnesium atom loses two electrons to get Neon's configuration, [He]2s22p6.
* Each Chlorine atom gains one electron to get Argon's configuration, [Ne]3s23p6.
* Thus formed Mg2+ and two Cl- ions combine together by forming MgCl2.
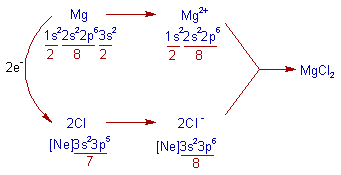
* The electrovalency of Mg is 2 and that of Cl is 1.
Note: The electronegativity difference between Mg (1.2) and Cl (3.0) is 1.8. Hence they form ionic bond.
3) AlF3 (Aluminium Fluoride) :
* The Aluminium atom loses 3 electrons and each Fluorine atom gains 1 electron to get their nearest inert gas: Neon's configuration, [He]2s22p6.
* Thus formed Al3+ and three F- ions combine together to give AlF3.
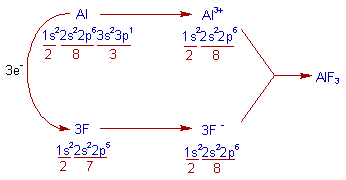
* The electrovalency of Al is 3 and that of F is 1.
Note: The bond between Al (e.n = 1.5) and F (e.n = 4.0) is ionic since the electronegativity difference between them is 2.5.
4) Na2O (Sodium monoxide) :
* In the formation of Sodium monoxide, the each sodium atom loses 1 electron and oxygen gains 2 electrons. While doing so, both atoms will get the nearest inert gas: Neon's configuration, [He]2s22p6.
* Thus formed two Na+ and O2- ions combine to form Na2O.
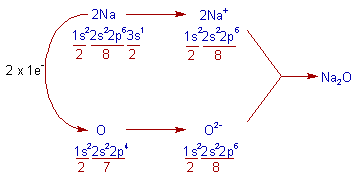
* The electrovalency of 'Na' is 1 and that of 'O' is 2.
Note: The bond between Na (e.n = 0.9) and O (e.n = 3.5) is ionic since the electronegativity difference between them is 2.6.
Author: Aditya vardhan Vutturi