1) It is a second order reaction.
2) Half life is independent of initial concentration of A.
3) The unit of the specific rate, k is L mol-1 s-1.
4) Half life is inversely related to the initial concentration of A.
Logic:
Except one, the remaining three statements are correct regarding the given reaction. The logic is to find out the order of reaction comparing the rates at different concentrations.
The rate law for a given reaction reaction can be written as follows:
![]()
where:
k = specific rate
[A] = concentration of reactant, A
n = order of reaction
Let
the initial rate: r1 = k[A1]n
the rate after doubling the conc. of A: r2 = k[A2]n
Also we know that:
[A2] = 2[A1] (since the concentration of A is doubled)
and
r2 = 4r1 (since the rate is increased by four times)
Now by comparing rates at two different conditions, it is possible to find the order of the reaction.
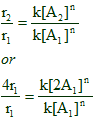
or
4 = 2n
Therefore:
n = 2
That means given reaction is a second order reaction.
* Now we know that for a second order reaction, the half life is inversely proportional to the initial concentration of the reactants and the units of specific rate can be written as: L mol-1 s-1.
In general:
![]()
unit for rate constant or specific rate, k is mol1-n Ln-1 time-1.
Conclusion:
Since it is a second order reaction, options 1,3 and 4 are correct statements.
1) What should be the order of reaction if the half life is independent of initial concentration of reactants?
2) What are the units of zero, first and third order reactions?
3) What will be the order of reaction if the rate is doubled when concentration of reactant is doubled?