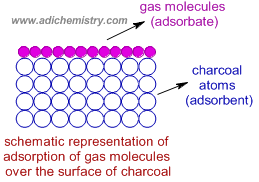
Adsorption is the accumulation or concentration of liquid or gas molecules (adsorbate) over the surface of a solid or a liquid (adsorbent). It is a surface phenomenon.
Adsorbate: The gas or liquid that is accumulated over the surface of a liquid or solid is known as adsorbate.
Adsorbent: The solid or liquid on whose surface, molecules of other substance are adsorbed. Solids, particularly in finely divided state, have large surface area and therefore act as good adsorbents.
E.g. activated charcoal, silica gel, alumina gel, clay, colloids, metals in finely divided state, etc.
Illustration: If a gas is taken in a closed vessel containing finely powdered charcoal, it is observed that the pressure of the gas in the enclosed vessel decreases. It is due to accumulation of gas molecules over the surface of charcoal.
Note: The charcoal is acting as adsorbent and gas as adsorbate.

Desorption: The reverse of adsorption is called as desorption.
The surface atoms or molecules of adsorbent are relatively unstable due to positive surface free energy. Unlike in the bulk, there are unbalanced residual forces at the surface as the molecules at the surface are not symmetrically surrounded by other molecules. Hence they have tendency to attract adsorbate molecules and retain them to minimize the surface energy.
In general, adsorption is an exothermic process (i.e., ΔH = -ve) i.e., heat is liberated since new bonds are formed. However entropy of the system is also decreased (i.e., ΔSsys = -ve) due to decrease in the number of microstates and decrease in the freedom of movement of molecules. Hence adsorption is thermodynamically more favourable at low temperatures.
Note: The value of ΔG becomes negative, only at low temperatures, when both ΔH and ΔSsys are negative.
Therefore, in general, at higher temperatures, the bonds between adsorbate and adsorbent are weakened and the reverse of adsorption i.e. desorption is favored.
It is important to note that above discussion is valid in most of the cases, especially for physical adsorption. However, there are few cases of chemical adsorption in which the bond dissociation energy of adsorbate molecules is greater than bond formation with the surface of adsorbent and thus results in net absorption of heat i.e. endothermic.
Adsorption is different from absorption.
Absorption involves the diffusion of molecules of a substance into the bulk of liquid or solid to form a solution.
E.g., Hydrogen gas is absorbed into the bulk of palladium metal, which is also known as occlusion.
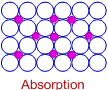
Absorption is a bulk phenomenon, whereas adsorption is a surface phenomenon.
Sorption: If both adsorption and absorption occurs simultaneously, the process is called as sorption.
Sorption = Adsorption + Absorption
Depending on the type of attractions between adsorbate and adsorbent, the adsorption can be divided into two types.
If the adsorbate molecules are attracted by weak van der Waals forces towards the adsorbent molecules, the adsorption is known as physical adsorption or physisorption.
Characteristics of physisorption:
* Energetics & kinetics: Physisorption is an exothermic process. However it is characterized by low enthalpy values (20– 40 kJ mol-1), due to weak van der Waals forces of attraction.
The activation energy for physisorption is also very low and hence it is practically a reversible process.
* Effect of temperature: Since physical adsorption is an exothermic process, it occurs more readily at lower temperatures and decreases with increase in temperature (Le-Chatelier's Principle).
* Effect of pressure: In case of physisorption of gases over solids, the extent of adsorption increases with increase in pressure as the volume of the gases decrease during adsorption (Le-Chatelier's Principle).
 |
During adsorption, the volume of gases decreases along with the liberation of heat. Hence at high pressures and low temperatures, adsorption is favored. Whereas at low pressures and high temperatures, desorption is favored. |
* Specificity: Since the van der Waals’ forces are universal, a given surface of an adsorbent does not show any preference for an adsorbate in physisorption i.e. it is not specific with respect to adsorbent.
* Nature of adsorbate: However, the extent of adsorption depends on the nature of gas (adsorbate). In general, easily liquefiable gases with higher critical temperatures) are readily adsorbed as the van der Waals’ forces are stronger, especially, near the critical temperatures.
E.g. Sulphur dioxide (critical temperature 630K) is adsorbed more than methane (critical temperature 190K) over activated charcoal under given set of conditions.
* Surface area of adsorbent: The extent of adsorption increases with the increase of surface area of the adsorbent. Hence finely powdered metals and porous substances having large surface areas perform well as adsorbents.
If the adsorbate molecules are bound to the surface of adsorbent by chemical bonds, the adsorption is known as chemical adsorption or chemisorption.
Characteristics of chemisorption:
* Energetics & kinetics: Chemisorption is also an exothermic process and the enthalpy values are higher (80-240 kJ mol-1) as it involves formation of chemical bonds.
However, the activation energy for chemisorption is high and occurs slowly. Hence it is also called activated adsorption. It is practically irreversible.
* Effect of temperature: Even though chemical adsorption is an exothermic process, it does not occurs slowly at lower temperature due to high kinetic energy barrier. Hence, like most chemical changes, the extent of chemisorption increases with increase in temperature up to certain limit and then after that it starts decreasing.
It is also observed that, in some cases, physisorption of a gas adsorbed at low temperature may change into chemisorption at a high temperatures.
* Effect of pressure: The chemisorption is not appreciably affected by small changes in pressure. However, very high pressures are favourable for chemisorption.
* High specificity: Chemisorption is highly specific and occurs only if there is some possibility of chemical bonding between adsorbent and adsorbate.
E.g. Oxygen is adsorbed on metals due to formation of oxide.
* Surface area: Like physisorption, chemisorption also increases with increase of surface area of the adsorbent.
1) Which of the following statements is true ? (GUJARAT SET SEPTEMBER 2018)
(A) Adsorption is always exothermic
(B) There is no heat change during adsorption
(C) Adsorption is always endothermic
(D) Adsorption can be exothermic or endothermic
2) Which one among the following is an incorrect statement about Van der Waals adsorption? (KERALA SET 2010)
A) Characterized by low heat of adsorption
B) Reversible
C) Decreases with increase in temperature
D) Activation energy of desorption is very high
1) D 2) D