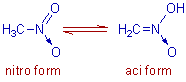Isomerism is the phenomenon of exhibiting two or more compounds with same molecular formula but different physical and chemical properties.
Isomers are the compounds showing different physical and chemical properties but same molecular formula.
The isomerism in organic compounds can be classified broadly into:
STRUCTURAL ISOMERISM
The structural isomerism or constitutional isomerism arises when atoms within a molecule are arranged in different orders. The structural isomers have the same molecular formula but different structural formulae.
The structural isomers usually show different physical and chemical properties.
The structural isomerism is further divided into:
CHAIN ISOMERISM
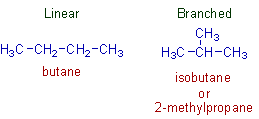
The chain isomerism arises due to different arrangements of carbon atoms leading to linear and branched chains. The chain isomers have same molecular formula but different types of chains i.e., linear and branched.
The chain isomers have almost similar chemical properties but different physical properties. For example, the branched chain isomers have lower boiling points than that of their linear counterparts. It is because, the linear ones have more surface area of contact and hence the intermolecular forces of attraction are maximum.
E.g. Two chain isomers possible with the molecular formula, C4H12.
i.e.,
n-butane: a linear chain isomer.
isobutane (or 2-methylpropane): a branched isomer
POSITIONAL ISOMERISM
The positional isomerism arises due to different positions of side chains, substituents, functional groups, double bonds, triple bonds etc., on the parent chain.
E.g.
1) Propyl chloride and isopropyl chloride are the positional isomers with the molecular formula, C3H7Cl. These isomers arise due to difference in the position of the chloro group on the main chain.
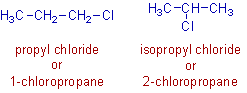
2) In the following positional isomers i.e., but-1-ene and but-2-ene the position of double is different but they have same molecular formula, C4H8.
![]()
3) The functional group, -OH is at different positions on the main chain of the following alcohols. They have the same molecular formula, C3H7OH.
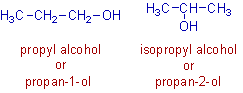
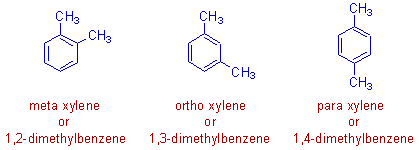
4) In the following positional isomers i.e., ortho xylene, meta xylene and para xylene, the relative positions of methyl groups on the benzene ring are different
FUNCTIONAL ISOMERISM
The functional isomerism arises due to presence of different functional groups. The functional isomers have the same molecular formula but possess different functional groups.
The functional isomers show different physical as well as chemical properties.
E.g.
1) The dimethyl ether is the functional isomer of ethyl alcohol. Both have the same molecular formula, C2H6O. However they have different functional groups.
![]()
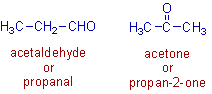
2) Acetaldehyde is a functional isomer of acetone. They have the same molecular formula, C3H6O but different functional groups. Acetaldehyde contains aldehyde, -CHO functional group, whereas acetone has ketone, >C=O functional group.
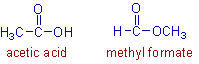
3) Both Acetic acid and methyl formate are also functional isomers. They have the same molecular formula, C2H4O2. Acetic acid is a carboxylic acid with -COOH group whereas methyl formate is an ester with -COOCH3 group.
METAMERISM
The metamerism arises when different alkyl groups are attached to same functional group.
E.g.
1) The following metamers contain the ether functional group. However they differ by the nature of alkyl groups attached to the oxygen atom.
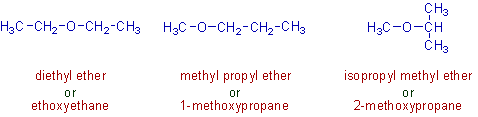
2) The metamerism is also possible in amines as shown below.
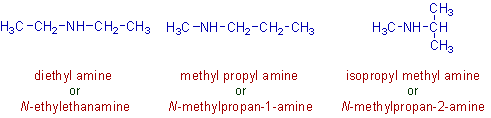
TAUTOMERISM
Tautomerism refers to the dynamic equilibrium between two compounds with same molecular formula. It is also called as desmotropism or kryptotropism or prototropy or allelotropism.
It is most often a special case of functional group isomerism. In general, the tautomers have different functional groups and exist in dynamic equilibrium with each other due to a rapid interconversion from one form to another.
It is very important to note that the tautomers are not the resonance structures of same compound.
1) Keto-enol tautomerism: The carbonyl compounds containing at least one α-hydrogen atom exhibit keto-enol tautomerism. The carbonyl group can be converted to enol form due to transfer of one of the α-hydrogen onto the oxygen atom.
Note: An alcoholic group on C=C is called enol. It is an alkene alcohol.
The % composition of keto enol tautomeric mixture depends on the relative stabilities of these two forms. In general the keto form is the low energy form and is more stable than the enol form. However the enol form is also stable in certain cases due to other stability factors.
The dynamic equilibrium between a keto form and an enol form exhibited by acetone is shown below. The tautomers are formed due to complete transfer of hydrogen. However the relatively stable keto form exists in higher percentage (about 99%).
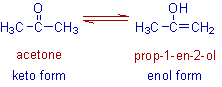
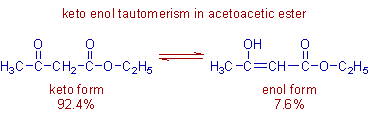
However enol form is also appreciably stable in case of 1,3-dicarbonyl compounds like acetoacetic ester (ethyl aceto acetic ester) as depicted below.
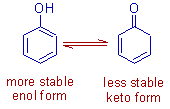
The enol form is more stable than the keto form in case of phenols due to resonance stabilization of aromatic ring.
2) The nitro-aci tautomerism: It is exhibited by nitro compounds containing atleast one α-hydrogen. In this case also, the hydrogen atom is transferred completely from one atom to another during the conversion of nitro form to aci form and vice versa.
E.g. The nitro-aci tautomerism exhibited by nitromethane is shown below.