The Wurtz reaction involves the coupling of two molecules of alkyl halides in the presence of sodium metal in anhydrous ether (or other unreactive polar aprotic solvents like tetrahydrofuran (THF), dioxane) at a temperature between 0 and 100 oC to form a new carbon–carbon bond, resulting in the formation of a symmetrical alkane. It is named after its discoverer, Charles Adolphe Wurtz, who first reported it in 1855.
| Dry Ether | ||
| R-X + 2Na + X-R | -----------------> | R-R + 2NaX |
Where X = halogen (Cl, Br, I)
IMPORTANT CONDITIONS FOR WURTZ REACTION
* The Wurtz reaction must be performed under anhydrous conditions because,
i) Sodium metal reacts violently with water and
ii) the alkyl free radical formed (see the mechanism below) during the reaction is highly reactive due to strong basic nature and therefore can abstract a proton from water.
* In case of alkyl and aryl fluorides as well as aryl chlorides, tetrahydrofuran is used as solvent instead of ether.
LIMITATIONS OF WURTZ REACTION
* The Wurtz reaction is limited to synthesis of symmetrical alkanes with even number of carbon atoms only.
E.g. Symmetrical alkanes like ethane, butane, hexane etc. with even number of carbon atoms can be prepared. But it is not possible to prepare alkanes with odd number of carbon atoms like propane, pentane etc.
The number of carbons in the alkane is always double that of alkyl halide (n ---> 2n type reaction).
* The Wurtz reaction is not suitable for the synthesis of alkanes with more than 12 carbon atoms.
* If two different alky halides are used, a mixture of alkanes is formed. It is usually difficult to separate the mixture and hence wurtz reaction not a suitable method to synthesize unsymmetrical alkanes.
E.g. The Wurtz reaction between R-X and R'-X yields not only R-R' but also R-R and R'-R'. This mixture cannot be separated easily.
* Methane cannot be prepared by this method.
* It is not compatible with functional groups that are sensitive to strong bases, such as aldehydes, ketones, and esters.
* When subjected to Wurtz reaction conditions, the vicinal dihalides yield alkenes, whereas geminal dihalides give alkynes.
Note: A modification of this reaction involving alkyl and aryl halides is called Wurtz-Fittig reaction. If only aryl halides are subjected to coupling, the reaction is called as Fittig reaction.
MECHANISM OF WURTZ REACTION
Step-1 : Formation of Alkyl Free Radical
* Initially one electron is transferred from sodium atom to alkyl halide resulting in the formation of an alkyl free radical.
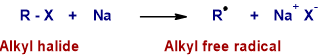
Step-2 : Formation of Carbonium ion
* One more electron is transferred from sodium atom to the alkyl free radical to give an alkyl cabanion.
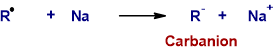
Step-3 : Nucleophilic substitution (SN2 Step)
* Thus formed alkyl anion displaces halide ion from the second molecule of alkyl halide. It is an SN2 reaction.
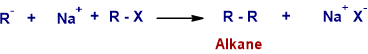
Side Reactions:
Elimination side reactions leading to alkenes are also possible, especially with bulky alkyl groups, which require more activation energy during the nucleophilic substitution (SN2) step due to steric hindrance.
WURTZ REACTION EXAMPLES
1) Ethane is formed when methyl chloride is treated with sodium metal in dry ether.
| Dry Ether | ||
| CH3-Cl + 2Na + Cl-CH3 | -----------------> | CH3-CH3 + 2NaCl |
2) Strained carbon skeletons like bicyclobutane ( bicyclo[1.1.0]butane ) can be prepared by an intramolecular Wurtz reaction of 1-bromo-3-chlorocyclobutane as shown below.
![preparation of bicyclo[1.1.0]butane by wurtz reaction](reaction-1.png)
3) When tert-butylhalides are subjected to Wurtz reaction, isobutylene is formed as the major product. It is because the elimination is favored over SN2 mechanism. The SN2 step requires more activation energy due to steric hindrance.
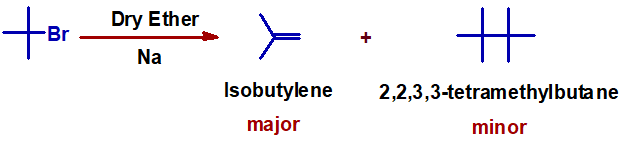
PRACTICE MCQ QUESTIONS FOR IIT JEE - NEET EXAMS
1) Which of the following alkyl halides gives the poorest yield of alkane in the Wurtz reaction?
A) CH3Cl
B) C2H5Br
C) (CH3)3CCl
D) n-C3H7Cl
2) When a mixture of CH3Br and C2H5Br is subjected to Wurtz reaction, how many different alkanes are formed?
A) 1
B) 2
C) 3
D) 4
3) Which of the following alkyl halides will predominantly give an alkene instead of an alkane in the Wurtz reaction?
A) CH3Cl
B) C2H5Br
C) (CH3)3CBr
D) n-C3H7Cl
4) Which of the following alkanes cannot be prepared by Wurtz reaction?
A) Butane
B) Methane
C) Hexane
D) All of the above
5) The Wurtz reaction must be carried out under anhydrous conditions because:
A) Alkanes are unstable in water.
B) Sodium reacts violently with water.
C) Alkyl halides are unstable in water.
D) Sodium becomes inert in presence of water.
6) Which of the following compounds can be best prepared by Wurtz reaction?
A) Methane
B) n-Pentane
C) Propane
D) n-Butane
7) The reagent used in the Wurtz reaction is:
A) Sodium (Na) metal in liq. NH3
B) Sodium (Na) metal in dry ether
C) Zinc (Zn) metal in dry ether
D) Sodium metal in water
8) The Wurtz reaction involves the coupling of:
A) Two alcohol molecules
B) Two alkyl halide molecules
C) Two aldehyde molecules
D) Two alkene molecules
9) The product(s) formed when a mixture of CH3Cl (Methyl Chloride) and C2H5Cl (Ethyl chloride) are subjected to Wurtz reaction is/are:
A) Only Propane
B) Only Ethane
C) Propane + Ethane + Butane
D) Propane + Butane
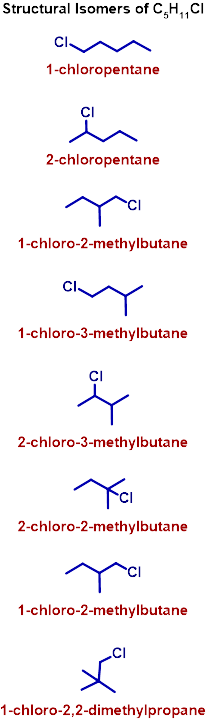
10) 2,2,5,5-tetramethylhexane is formed when an alkyl halide with formula C5H11Cl undergoes Wurtz reaction. The alkyl halide is:
A) 2-chloropentane
B) 2-chloro-3-methylbutane
C) 1-chloro-2-methylbutane
D) 1-chloro-2,2-dimethylpropane
Hint: The possible structural isomers for the formula C5H11Cl are given below. Also keep in mind that a new C-C bond is formed.
Answers:
1) C
2) C
3) C
4) B
5) B
6) D
7) B
8) B
9) C
10) D