* The organomagnesium halides are known as Grignard reagents. These are extremely important reagents developed by the French chemist Francois Auguste Victor Grignard, who was awarded the Nobel Prize in 1912 in Chemistry for this work.
The Grignard reagent is represented as R-Mg-X, where
R = alkyl / aryl / alkenyl / allyl group
X = Cl / Br / I
* The reactions involving Grignard reagents, as sources of nucleophiles, are usually referred to as Grignard reactions.
3) Reactions of Grignard reagent
PREPARATION OF GRIGNARD REAGENT
* The Grignard reagents are prepared by the action of activated magnesium (Rieke magnesium) on organic halides in suitable solvents like Diethyl ether, Et2O or Tetrahydrofuran, THF in anhydrous conditions.
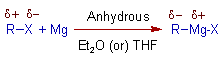
* This is an oxidative insertion of magnesium between carbon and halogen bond, which involves oxidation of Mg(0) to Mg(II). The mechanism of this reaction if not quiet conclusive.
* The Grignard reagents are in equilibrium with the dialkylmagnesium species R2Mg and MgX2 (Schlenk equilibrium).
![]()
* In the formation of Grignard reagent, the polarity of carbon attached to the halide group is reversed. This reversal in polarity is called as umpolung.
REACTION CONDITIONS
Activation of magnesium metal:
* Magnesium metal is usually unreactive due to formation of oxide layer on its surface. Hence it should be activated by dislodging this layer. It is achieved by adding small amount of iodine or 1,2-dioiodoethane or by using ultrasonic sound.
This problem can also be obviated by using Rieke magnesium, which is in the form of highly reactive small particles of magnesium with large surface area. It is prepared by reducing MgCl2 with lithium metal.
Solvent:
* Ether solvents like Diethyl ether, Et2O or Tetrahydrofuran, THF or Dimethoxyethane, DME or Dioxane are most suitable for the preparation of Grignard reagents. It is because they are not only unreactive with magnesium but also dissolve and stabilize the Grignard reagents by forming Lewi's acid base complexes.
* The major disadvantage of Grignard reagents is they react with protic compounds like water, alcohols, thiols etc. Hence the reaction must be carried out under anhydrous conditions avoiding moisture.
* These reagents must not be exposed to air as they also react with oxygen by giving peroxide species which are converted to corresponding alcohols during hydrolytic workup. To avoid this, it may be preferable to carry out the reaction in nitrogen or argon atmosphere.
![]()
PREPARATION OF DIFFERENT TYPES OF GRIGNARD REAGENTS
* The alkyl Grignard reagents are prepared from the corresponding chlorides or bromides or iodides. The order of reactivity of alkyl halides with magnesium is RCl < RBr < RI. Alkyl fluorides are seldom used due to much less reactivity.
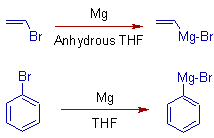
* The alkenyl and phenyl Grignard reagents are prepared from the corresponding bromides or iodides in more effective co-ordinating solvent like THF.
E.g. Vinyl bromide and bromobenzene can be converted to corresponding Grignard reagents by reacting them with magnesium metal in anhydrous THF.
* The alkynyl Grignard reagents are prepared by deprotonating 1-alkynes with another Grignard reagent like Ethylmagnesium bromide.
E.g. Propyne can be deprotonated with ethylmagnesium bromide to give propynylmagnesium bromide.
![]()
* The allylic Grignard reagents may undergo coupling reactions. Hence they are generated in situ whenever required in the Grignard reactions.
* Grignard reagents can also be prepared by transmetallation.
E.g. Alkyllithiums can give Grignard reagents when treated with magnesium salts.
![]()
REACTIONS OF GRIGNARD REAGENTS
* The Grignard reagents are highly basic and can react with protic compounds like water, acids, alcohols, 1-alkynes etc., by giving corresponding alkanes.
E.g. Ethylmagnesium bromide liberates ethane gas when treated with water.
![]()
The reaction of Grignard reagent with D2O can be used to introduce a deuterium atom selectively at a particular carbon atom.
![]()
* However the Grignard reagents are less basic than organolithiums and hence are more suitable nucleophiles for carbon-carbon bond formation.
* The Grignard reagents are used as sources of carbon nucleophiles (carbanions) and can react with electrophilic centers. The addition reactions involving Grignard reagents with compounds containing polarized multiple bonds like aldehydes, ketones, esters, acid halides, nitriles, carbon dioxide etc., are termed as Grignard reactions.
* The reactivity of carbonyl compounds with Grignard reagents follow the order: aldehydes > ketones > esters > amides
MECHANISM OF GRIGNARD REACTION
* The first step in the Grignard reaction is the nucleophilic addition of Grignard reagent to the polar multiple bond to give an adduct which upon hydrolytic workup gives the final product like alcohol.
E.g. The mechanism of reaction with a carbonyl compound is shown below.
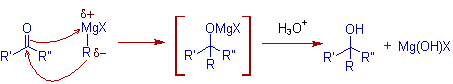
APPLICATIONS OF GRIGNARD REAGENT
Following is the summary chart of applications of Grignard reagent in modern organic synthesis.
| Grignard reaction | Product | ||
|
R-Mg-X + |
Formaldehyde ( HCHO ) |
-------> |
A primary alcohol: R-CH2-OH |
| Aldehyde (R'-CHO) |
-------> |
A secondary alcohol: R'-CH(OH)-R | |
| Ketone (R'-CO-R") |
-------> |
A tertiary alcohol: R'-CR"(OH)-R | |
| Ester (R'-COOR") |
-------> |
A tertiary alcohol: R'-CR(OH)-R | |
| Acid halide (R'-COX) |
-------> |
A tertiary alcohol: R'-CR(OH)-R | |
| CO2 |
-------> |
A carboxylic acid: R-COOH | |
| CS2 |
-------> |
A dithionic acid: R-CSSH | |
| SO2 |
-------> |
A sulphinic acid: R-SOOH | |
| SO3 |
-------> |
A sulphonic acid: R-SO2OH | |
| nitriles (R'-CN) |
-------> |
A ketone: RCOR' | |
| Hydrogen Cyanide (HCN) |
-------> |
An aldehyde: RCHO | |
| Oxiranes (epoxides) |
-------> |
Alcohols | |
| Weinreb amide |
-------> |
A ketone | |
| cyanogen |
-------> |
A nitrile | |
| choramine |
-------> |
An amine | |
| Iodine |
-------> |
Alkyl iodide | |
| Sulfur |
-------> |
A thiol | |
| halides of B, Si, P, Sn |
-------> |
compounds with C- hetero atom bonds | |
| CdCl2 | -------> | Dialkyl cadmium | |
1) The addition of Grignard reagents to formaldehyde furnishes primary alcohols.
E.g. The addition of Ethylmagnesium iodide to formaldehyde followed by hydrolytic workup furnishes Propyl alcohol, a primary alcohol.
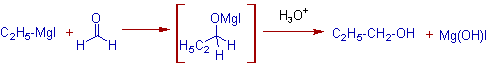
2) The Grignard reaction with aldehydes other than formaldehyde gives secondary alcohols.
E.g. The addition of Methylmagnesium iodide to acetaldehyde gives Isopropyl alcohol.
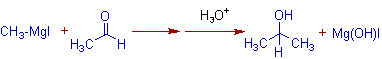
3) The addition of Grignard reagent to ketones furnishes tertiary alcohols.
E.g. The addition of Methylmagnesium iodide to acetone gives tert-Butyl alcohol.
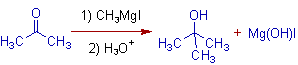
Stereochemistry:
The carbonyl carbon of an unsymmetrical ketone is a prochiral center. Therefore the addition of a Grignard reagent can take place on either face of the carbonyl group with equal chance. Hence a racemic mixture is formed in absence of asymmetric induction.
E.g.
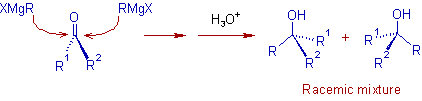
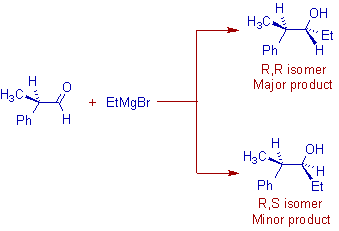
However a mixture of diastereomers is formed when the ketone or aldehyde contains at least one chiral center. The predominant stereoisomer formed in this case can be predicted by using Cram's rule.
E.g. The reaction of (R)-2-phenylpropanal with ethylmagnesium bromide, an achiral Grignard reagent furnishes the (R,R)-2-phenyl-3-pentanol as major product.
Side reactions:
However, the abstraction of an α-hydrogen by Grignard reagent (in this case it acts as a base) is observed with sterically hindered ketones to furnish an enolate intermediate. The protic workup of the enolate ends up in the recovery of the starting ketone.
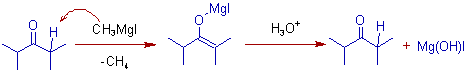
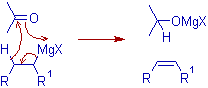
If the Grignard reagent contains a β-hydrogen, reduction of carbonyl compound by hydride transfer may compete with the desired addition reaction (see below). Hence the Grignard reagent with smallest possible alkyl group is to be used to avoid this side reaction. Also the use of corresponding organolithium compounds is advisable to suppress the enolization products.
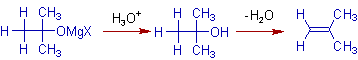
It is also observed that the tertiary magnesium alkoxides bearing a β-hydrogen, may undergo a dehydration reaction during protic workup, and thus by giving an elimination product, alkene instead of alcohol.
E.g.
4) The esters are less reactive than aldehydes and ketones. However they give tertiary alcohols with excess (2 moles) of Grignard reagent. The initial addition product formed will decompose to a ketone which reacts with the second Grignard reagent to furnish the tertiary alcohol finally.
E.g. Ethyl acetate reacts with two moles of phenylmagnesium bromide and thus by furnishing 1,1-diphenylethanol, a tertiary alcohol.
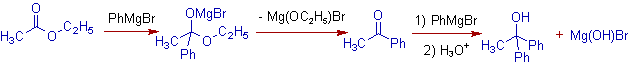
5) The acid halides also react with 2 moles of Grignard reagent to furnish tertiary alcohols. Again the reaction proceeds through the intermediate ketone.
E.g. Acetyl chloride reacts with two moles of Ethylmagnesium bromide to furnish 3-methylpentan-3-ol.
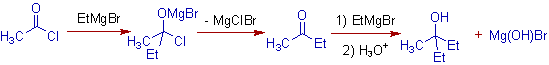
However, it is also possible to get the ketone in higher yields by using one mole of Grignard reagent.
6) The Grignard reagents react with carbon dioxide to give carboxylic acids.
E.g. Methylmagnesium chloride gives acetic acid when reacted with carbon dioixide.
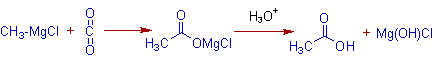
An analogous reaction of Grignard reagent is observed with carbon disulphide, CS2, to give alkanedithionic acid.
E.g. Ethanedithionic acid can be prepared by reacting methylmagnesium chloride with carbon disulphide, CS2.
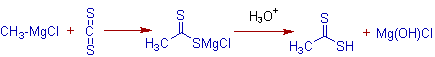
Also in another analogous reaction with sulfur dioxide, SO2, an alkanesulphinic acid is formed.
E.g. Methanesulphinic acid is formed when methylmagnesium chloride reacts with sulfur dioxide, SO2.
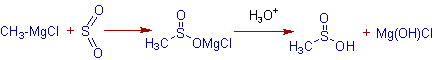
Whereas, alkane sulphonic acids are formed with sulfur trioxide, SO3.
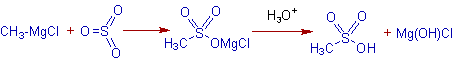
7) The nitriles furnishes ketones with Grignard reagents.
E.g. Acetonitrile gives acetone when reacted with methyl magnesium iodide.
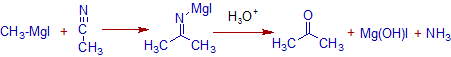
However, aldehydes are obtained when Grignard reagent are added with hydrogen cyanide, HCN followed by hydrolysis.
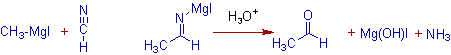
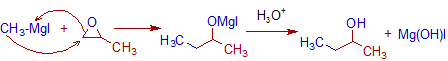
8) The oxiranes (epoxides) furnish alcohols with Grignard reagents.
E.g. Secondary butyl alcohol is obtained when 2-methyloxirane reacts with methylmagnesium iodide.
The less substituted carbon of oxirane is substituted by the alkyl group of Grignard reagent.
9) Addition of an N-methoxy-N-methyl amide, also known as Weinreb amide, RCON(Me)OMe, to the Grignard reagent gives a ketone. Initially the Grignard reagent is added to the Weinreb amide, which further undergoes hydrolysis to furnish ketone.
E.g. The addition of n-butylmagnesium bromide to the following Weinreb amide furnishes 3-heptanone.
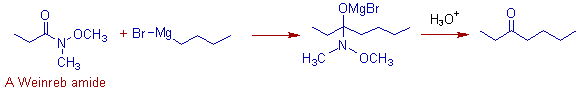
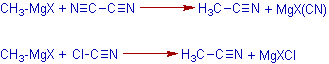
10) The Grignard reagents are also used to prepare nitriles by reacting them with cyanogen or cyanogen chloride.
11) Amines can be prepared by reacting these reagents with Chloramine, NH2Cl.
![]()
12) The alkyl iodides can be prepared via Grignard reagents. The alkylmagnesium chlorides or bromides are treated with Iodine to get corresponding alkyl iodides.
![]()
13) A Wurtz like coupling reaction is also possible when the Grignard reagent is treated with an alkyl halide to furnish an alkane. Indeed it is a side reaction that may be possible during the preparation of Grignard reagent. This reaction is catalyzed by Cuprous (CuI) ions.
![]()
14) Just like oxygen, the sulfur atom is also inserted into the Grignard reagent, which gives a thiol upon protic workup.
![]()
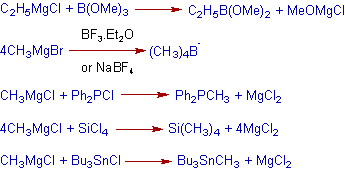
15) The Grignard reagent is also used in the making of bond between a carbon and other hetero atom like B, Si, P, Sn etc. These applications are depicted in the following reactions.
16) Dialkyl cadmium compounds are formed when the Grignard reagents are made to react with cadmium chloride.
![]()
The dialkyl cadmium compounds furnish ketones upon reacting with acid halides.
![]()
GRIGNARD REACTION - MCQ - IIT JEE - NEET - JAM - GATE - CSIR NET & SET EXAMS
1) (CH3)3CMgBr on reaction with D2O produces : (GUJARAT SET DEC 2002)
(A) (CH3)3CD
(B) (CH3)3COD
(C) (CD3)3CD
(D) (CD3)3COH
2) Which of the following compounds will react with isopropyl magnesium bromide followed by water to give 2, 4-dimethyl pentan-2-ol ? (GUJARAT SET DEC 2002)
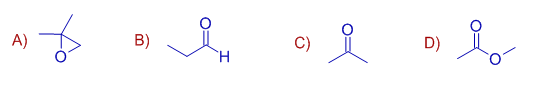
3) Reaction of methyl benzoate with CH3MgBr generates : (GUJARAT SET DEC 2004)
(A) α, α - Dimethyl benzyl alcohol
(B) Acetophenone
(C) Methyl-o-methyl benzoate
(D) Benzyl alcohol
4) Choose the correct statement regarding the following reaction.: (WEST BENGAL SET 2014 CHEMISTRY)
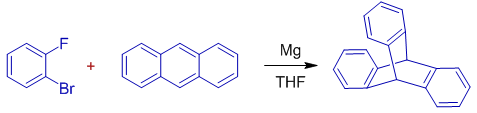
(A) A Grignard reagent from the dihalobenzene adds to anthracene, followed by nucleophilic displacement of fluoride anion to form the product.
(B) Magnesium reduces anthracene to a reactive dianion that bonds to the dihalobenzene.
(C) A Grignard reagent from the dihalobenzene metalates the anthracene, and this nucleophile adds to the remaining fluorobenzene.
(D) A Grignard reagent from the dihalobenzene decomposes to benzyne, which then cycloadds to anthracene.
5) Which one of the following reactions will not result in the formation of anisole? (GATE 1995)
A) Phenol + dimethyl sulphate in the presence of base
B) Sodium phenoxide treated with methyl iodide
C) Reaction of diazomethane with phenol
D) Reaction of methylmagnesium iodide with phenol
6) The conversion, PhCN ------> PhCOCH3, can be achieved most conveniently by reaction with: (GATE 1997)
A) CH3MgBr followed by hydrolysis
B) I2 – NaOH;CH3I
C) dil. H2SO4 followed by reaction with CH2N2
D) LAH followed by reaction with CH3I
KEY & EXPLANATION
1) A (see the mechanism) 2) A 3) A 4) D 5) D 6) A